What are clinical trials and just how important are they in cancer care?
Dr Huren Sivaraj , 2 November 2020
The field of oncology has advanced rapidly over the years, with the research and development of new drugs, drug combinations and treatment strategies for cancer.
Case in point: According to the IQVIA Global Oncology Trends 2019 report, there was a 19 per cent increase in the number of late-stage pipeline therapies between 2017 and 2018, along with a record of 15 new oncology therapeutic drugs launched for 17 indications in 2018. Within the pipeline and across all phases of clinical development, the most intense activity is focused on nearly 450 immunotherapies with more than 60 different mechanisms of action.
A total of 98 Next-Generation Biotherapeutics – defined as cell, gene and nucleotide therapies - are also under clinical investigation and leverage 18 different approaches. Gene therapy clinical trials worldwide are progressing as well. According to consumer data portal Statista, as of November 2019, 55 percent of clinical trials in cell and gene therapy development were in phase two, and 30 percent were in phase one of clinical trials.
These developments point to the potential for improved clinical outcomes for cancer patients, as they navigate a host of new options.
What is cancer?

Cancer refers to a large group of diseases caused by an uncontrolled division of abnormal cells in the body. These have the ability to spread into and destroy surrounding healthy tissue and organs. The latter process is called metastasising and is a major cause of death from cancer. Cancer can start nearly everywhere in the human body, and lead to growths known as tumours that can take the form of solid masses of tissue or blood-borne cancers known as leukemias. Such tumours, which are malignant, can also spread to other tissues and sometimes recur even after removal. On the other hand, benign tumours do not spread.
There are more than 100 types of cancer, which are named after the area where they originated. However, these are grouped into several broad categories: carcinomas, the most common type of cancer, begin in the skin or tissues that line organs; sarcoma form in bone and soft connective tissues, including muscles, cartilage and blood vessels; leukemia is a cancer of bone marrow, which creates vital blood cells, and lymphoma and myeloma are cancers of the immune system. Today, with a greater understanding of the molecular or genetic features of cancers, we have started to classify cancers beyond the typical organ of origin. We will, however, cover this in a separate article.
A genetic disease, cancer is caused by changes to genes that control the way our cells function, particularly how they grow and divide. These are known as DNA mutations, and can cause cells that should be replaced to survive, and new cells to form when they are not needed. With that being said, it can also be caused by environmental exposure to elements such as chemicals in tobacco smoke, radiation and ultra-violet rays as the driving force behind the earlier described genetic changes.
According to the World Health Organization (WHO), cancer is the second leading cause of mortality globally. It affects people across a spectrum of ages, races and ethnic groups, though its incidence rate varies from group to group. The disease is highly prevalent in those aged 50 and above. The WHO states that between 30 - 50 per cent of cancers can currently be prevented by avoiding risk factors and implementing existing evidence-based prevention strategies, while early detection and management of cancer patients can help to reduce mortality rates.
Why are clinical trials important?

While novel cancer therapies are being developed at an unprecedented rate, they typically undergo between 10 and 15 years of research and development before being approved for use by doctors. This protracted period usually involves a rigorous process of laboratory studies before researchers proceed to the final stage - human clinical trials.
Led by a principal investigator, clinical trials are crucial research studies that test the safety and effectiveness of new treatments on cancer patients on a volunteer basis. Cancer clinical trials are designed to test new ways to treat, diagnose and prevent cancer, while managing its symptoms and treatment side effects. Among the novel therapies tested in clinical trials, are new drugs, new approaches to surgery or radiation therapy, as well as new combinations of treatments or new methods such as gene therapy.
Clinical trials are carefully designed and reviewed, and have to be approved before they can take place. Typically, human biomedical clinical trials are carried out in four phases, from testing the safety of new drugs on a small group of people, to testing them in a wide population over a longer time frame.
Such trials can help to improve clinical outcomes - along with patients’ quality of life and chances of surviving the disease - by providing access to new treatments that are possibly more effective. For instance, recent studies in immunotherapy and precision medicine have resulted in improved outcomes in cancers of the lung, colon and melanoma.
As an example of the progress in expanding the role of immunotherapy through clinical trials, the Impassion 130 study, which was led by Professor Peter Schmid, clinical director of St Bartholomew’s Hospital’s Breast Cancer Centre in London, showed that combining chemotherapy with an immunotherapy drug Atezolizumab can help extend the lifespan of triple-negative breast cancer patients afflicted with the aggressive and difficult to treat cancer. This signalled significant progress in the field with the approval of immunotherapy in a specific group of breast cancers which are generally understood to be more aggressive.
The global study included patients from Singapore, and was first reported in 2018. The following year, the United States’ Federal Drug Authority approved the combination of the aforementioned drug with chemotherapy for the treatment of triple-negative, metastatic breast cancer in selected patients.
Are cancer clinical trials free?
Patients typically do not have to pay for the majority of clinical trial costs, as trial sponsors cover the costs of research and data analysis. Generally, newer treatments that are currently in the market are partially or fully sponsored. Other components such as routine doctor visits and investigations may also be sponsored. In Singapore, cancer clinical trial participants work closely with a clinical trial team including doctors, nurses and clinical trial coordinators. The latter will provide advice on any treatment costs involved, which may be covered by your insurance plan.
Myths about cancer clinical trials

While all clinical trials have risks, the overarching question is often whether their benefits will outweigh the risks. For this, patients should seek advice from their physicians to help them properly assess their landscape of options and thus make an informed decision. Nonetheless, there are common misconceptions about clinical trials that should be addressed:
Myth #1: Clinical trial patients are exposed to danger as guinea pigs
Patient safety is a top priority in all clinical trials, and patients are not exposed to unnecessary risks. It is important to note that therapies in clinical trials are adequately investigated for their safety in the laboratory before being tested on patients. Initial safety studies are typically conducted in a very small group of patients in the earliest phase, and strict monitoring guidelines within subsequent phases of research ensure that side effects are acknowledged and appropriately managed by clinical trial investigators.
Myth # 2: Cancer clinical trial candidates will get a placebo
A placebo is a pill, injection or treatment designed to have no therapeutic value or adverse side effects. These are used to assess a treatment’s effect on a particular condition. It is important to note that placebos are never used in place of standard care, and a patient enrolled in a clinical trial will either receive standard care or an experimental therapy. Furthermore, it is rare for experimental placebo therapies to be conducted in clinical trials, and typically only occurs when there is no standard treatment available for the particular cancer type or stage. If the trial design includes a placebo, this information must be made available to you in the informed consent form.
Myth #3: It is too difficult to get enrolled in a clinical trial
According to the WHO, more than 60,000 clinical trials were registered worldwide in 2018. All of these have different trial designs, objectives and eligibility criteria. The current trend in oncology research is to make the eligibility criteria more inclusive for a wider section of the cancer patient population. This means that there is a good chance you will match the eligibility criteria of a number of cancer clinical trials. Oncoshot’s profile building tool allows you to easily identify the clinical trials - among thousands of ongoing trials - with key eligibility criteria that match a specific cancer profile.
Myth #4: I won’t be able to leave a clinical trial I enrolled in
While having candidates complete their clinical trial is favourable for research as well as clinical outcomes, trial patients have the right to withdraw from the trial at any point, without having to justify their decision.
Myth #5: Cancer clinical trials are a last-ditch effort
Contrary to the belief, cancer clinical trials are not just for late-stage patients who have exhausted their treatment options. Cancer clinical trials are designed for various cancer types and stages, and trial options can improve clinical outcomes at different points of your treatment process.
How to access cancer clinical trials services

Today, there is a compendium of clinical trials available for all stages of cancer, globally. According to German statistics portal Statista, the number of clinical trial starts in oncology between 2000 and 2018 rose from 500 to 2,300. And WHO’s International Clinical Trials Registry Platform suggested that at least 2,800 new cancer treatment related trials were registered in 2017 alone.
However, while many new strategies are currently being studied across the world, it is still a challenge for even medical professionals to provide patients with access to active clinical trials. This is because sifting through a vast number of studies taking place globally involves a manual and time-consuming process that is daunting even for experienced oncologists.
Patients, caregivers and oncologists have limited tools to help identify suitable trials outside their site of care. In fact, most oncologists typically rely on word-of-mouth updates, on top of media reports, for information on new cancer clinical trials. For that reason, compounded by physicians’ own perceptions that enrolling patients in clinical trials is a time-consuming process, many primary caregivers may be less inclined to refer patients to clinical trials.
According to a study published in the American Society of Clinical Oncology Educational Book 36 (October 29, 2018), fewer than one in 20 adult patients with cancer enrol in cancer clinical trials.
The answer to solving this connundrum may lie in the appropriate use of technology. To improve the efficacy of oncology research as well as clinical outcomes, medical oncologists and pharmaceutical companies are increasingly turning to Big Data and Artificial Intelligence (AI). These technologies help to match cancer patients to the most suitable clinical trials in a matter of minutes. In fact, research by the Journal of the American Medical Informatics Association revealed that AI-based algorithms improved trial screening efficiency by more than 400 per cent.
Beyond matching cancer patients to the relevant clinical trials with greater speed and accuracy, AI-powered trial matching algorithms have the potential to create more diverse trial cohorts, thus helping to elevate oncology research. On a wider scale, it could even lead to more international trial collaboration.
How Oncoshot can help you
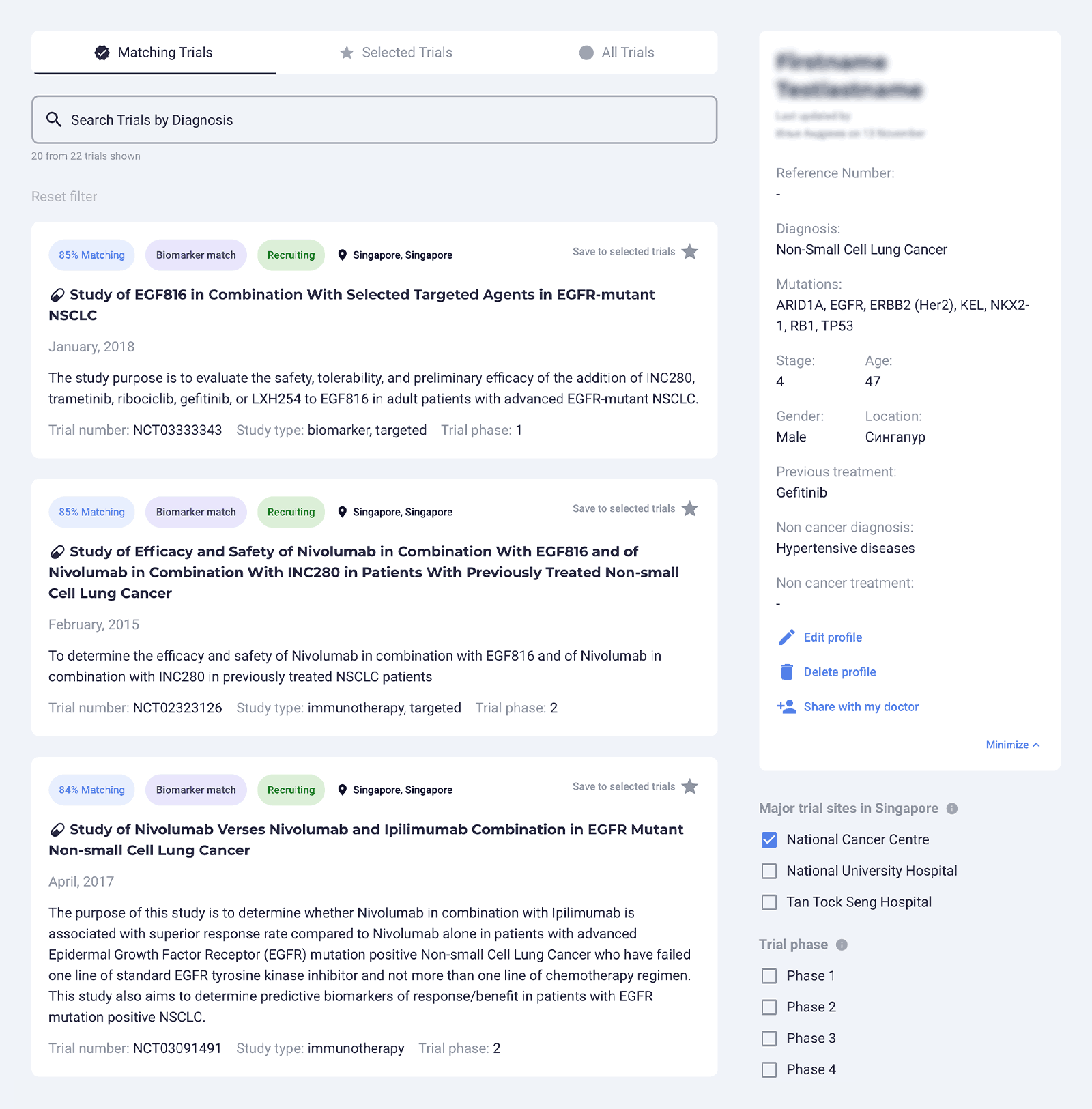 Image: Oncoshot presents cancer clinical trial matches for patients according to eligibility criteria
Image: Oncoshot presents cancer clinical trial matches for patients according to eligibility criteria
An example of an AI-powered platform that aims to improve the efficiency and efficacy of cancer clinical trials is Oncoshot. The patient-to-trial platform taps into a global database of more than 10,000 clinical trials, enabling the identification of suitable and promising trial options for patients in real time.
Driven by a complementary system that combines the expertise of AI and oncology specialists, Oncoshot parses critical information relevant to each patient’s unique medical condition to establish precise matches that take into account key trial eligibility criteria. The system’s accuracy is routinely assessed by a medical oncologist and clinical research assistant to ensure patients are matched to only the most relevant cancer clinical trials.
Sign up for free and create your profile on Oncoshot, where you can access ongoing cancer clinical trials taking place both locally and globally - whether you are a patient or oncologist. If you are a patient, you can then discuss these options with your primary physician, who can best counsel you on the feasibility of the trials and help you to get enrolled. The platform has an easy-to-use interface that includes details on the rationale behind each match, thus doing away with confusing manual searches.
Besides matching patients to trials and vice-versa, we also provide a concise list of cancer clinical trials that oncologists can access for free. They can choose to receive automatic updates on new cancer clinical trials by signing up here. Our company is working on building South-east Asia’s first ecosystem for cancer clinical trial and treatment options.